An article published in the journal “Nature” describes a new variant of the CRISPR-Cas9 genetic technique capable of obtaining more targeted modifications by inserting, eliminating and modifying pieces of DNA. A team from the Broad Institute of MIT and Harvard combined the CRISPR-Cas9 technique as it is currently used with a reverse transcriptase, in simple words an enzyme that can transcribe RNA into DNA. The result is what David Liu, one of the senior authors of the research, defined a greater flexibility in the target choice and greater precision in the modifications.
The CRISPR (clustered regularly interspaced short palindromic repeats) acronym refers to prokaryotic DNA segments containing short repeated sequences. The expression CRISPR/Cas refers to a prokaryotic immune system conferring a genetic resistance to foreign elements.
CRISPR-based techniques have already been refined over the years, now a further improvement has been announced. The new approach, which has been called “prime editing”, has been compared by David Liu to a word processing program with its search and replace tools, meaning it as a step forward with respect to the correction of a text written with a pencil. The goal is a greater precision in genetic modification operations to obtain greater flexibility and even higher safety by avoiding damage to the modified DNA.
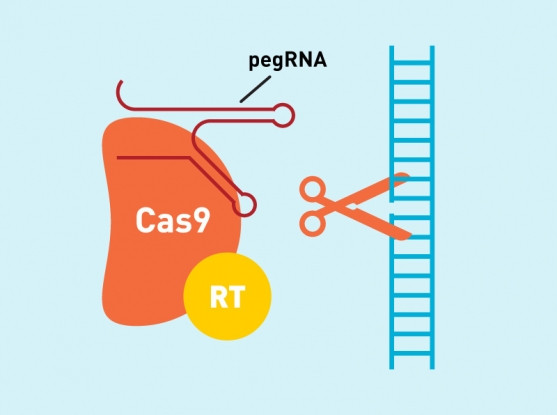
In prime editing, there’s the presence of an enzyme for reverse transcriptase that uses RNA to guide the insertion of new DNA sequences into the cells that are the target for the modification. For this reason, RNA has been called peg, the acronym for “prime editing guide”. PegRNA is specifically created to guide the modification to its target, where the Cas9 enzyme cuts a piece of DNA. PegRNA also contains additional RNA nucleotides to encode the new modified sequences, which are read by the reverse transcriptase (referred to as RT in the image) to encode the corresponding DNA nucleotides in the right place.
The researchers conducted experiments to eliminate genetic mutations that cause sickle cell anemia, which requires the conversion of a specific thymine to adenine, and Tay Sachs disease, which requires the removal of four DNA “letters” to a precise location in the genome. Efficiency has reached 55% and 35% respectively with very few side effects.
David Liu intends to continue optimizing the prime editing approach to maximize efficiency in many different types of cells. The use in genetic therapies is the main objective with the expansion of the experimentation already begun with the already existing CRISPR techniques.

